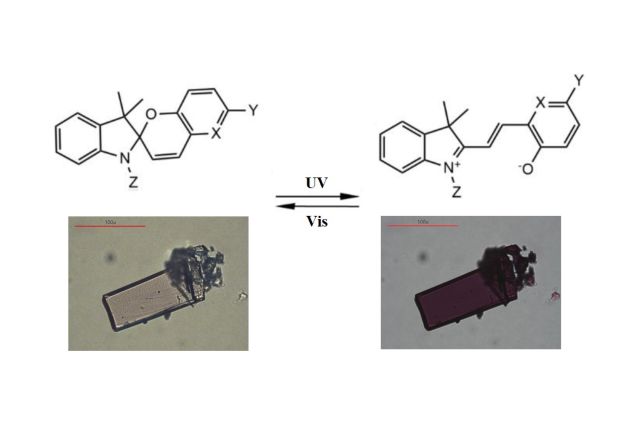
Spiropyran-merocyanine photochromic structural transformation studied in-situ in single-Micro/Nanocrystals by Electron Diffraction
Professor
UC Los Angeles
Area(s)
Characterization and Properties
Synthesis and Formulation